High prevalence of sexually transmitted disease is fostering the growth of the HPV and Pap testing market
HPV
and pap testing are used in detecting HPV infection, are often routine. It is
used to diagnose a sexually transmitted disease such as HIV, cervical cancer,
etc. HPV and pap testing are important not only to detect current infections
but also to detect potential risks for an individual's sexual behavior. This
test helps to reduce the risk of spreading infections to sexual partners and to
children who may be born and to individuals who may already have such diseases.
HPV infection is more common among men who know that they have it.
The
prevalence and incidence of cancer around the globe is relatively high and this
is a key factor driving the growth of the HPV and Pap testing market. According
to the American Cancer Society, cervical cancer is most frequently diagnosed in
women between the ages of 35 and 44 with the average age at diagnosis being
50. More than 20% of cases of cervical
cancer are found in women over 65. The cervical cancer death rate dropped
significantly with the increased use of the Pap test. Thus, the growing
prevalence of cervical cancer is fueling the growth of the HPV
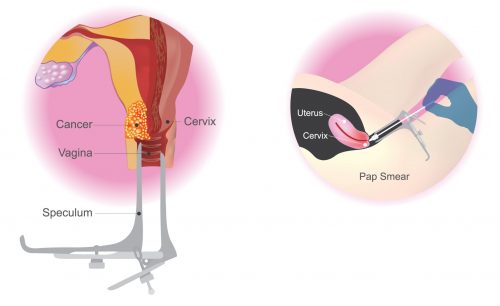
and Pap testing market. Pap test is a highly preferred test to detect the
cervical cancer which includes the microscopic observation of specimens.
North
America is projected to gain significant growth over the forecast period and
this is attributed to the implementation of strict guidelines for screening of
these tests by the government is again contributing to the growth of the
market. For instance, in August 2018, updated screening guidelines were released
by the United States Preventive Services Task Force. Women ages 21 through 29
should be screened with a Pap test in every 3 years. Women ages 30 through 65
should be screened with high-risk HPV testing alone in every 5 years, with Pap
and high-risk HPV testing in every 5 years, and a Pap test alone in every 3
years.
Key Developments:
1. In September 2020, Roche the
announced U.S. Food and Drug Administration (FDA) approval for the expanded use
of CINtec® PLUS Cytology, the first triage test based on biomarker technology
for women whose cervical cancer screening results are positive for high-risk
types of human papillomavirus (HPV)
2. In February 2018, BD (Becton,
Dickinson, and Company announced that it has received pre-market approval from
the U.S. Food and Drug Administration (FDA) for the BD Onclarity(TM) HPV assay.
The test detects 14 types of high-risk human papillomavirus (HPV) for cervical
cancer screening ("Pap test") in the BD SurePath(TM) liquid-based
cytology vial.


I am here to appreciate Dr OGU for using his herbal medicine to cure my Herpes virus. Is about 2 years and 6 months now I have been living with this virus and it has been a serious problem to me, I was so confused cause i have been taking several drugs to be cured but all of my effort was in vain,one morning i was browsing through the internet then i saw several testimonies about Dr. OGU curing people from Herpes virus and immediately i contacted Dr. on his email: drogugusolutionhome@gmail.com , i told him about my troubles and he told me that i must be cured, he gave me some instructions and which i rightly followed. so he prepared a herbal medicine and sent it to me via DHL which i used for 2 weeks and i was cured everything was like a dream to me and my Herpes virus was totally gone, dr .OGU , God bless you and give you more power and ability for more cures.i dont know if there is any one out there suffering for herpes virus or any of these diseases.. DIABETES, CANCER,GENITAL AND SIMPLEX HERPES,LOW SPERMS COUNT,SYPHILIS, HIV/AIDS, FIBROID,COPD,MENINGITIS,HEPATITIS A,B [HBV] DISEASES, CHLAMYDIA, .etc why don't you contact dr. Ogu today and be free from your diseases because he is very good and honest Doctor. contact him via email; drogugusolutionhome@gmail.com or text/call via: +1 (719) 629 0982
ReplyDelete